Introduction to Medical Device Regulatory & Biocompatibility Conferences
The Medical Device Conference 2026 will be the big day for the global MedTech industry. Manufacturers, service providers and regulators turn to global models for transparency, collaboration and compliance readiness as regulatory scrutiny intensifies and innovation cycles accelerate. The Medical Device Conference 2026 brings together global MedTech leaders to discuss biocompatibility, risk management, and regulatory safety in an increasingly integrated healthcare landscape.
This is the Medtech Forum 2026 and is one of the most relevant industry conferences of the year, as it addresses innovation, compliance, and growth.

In 2026, medical device manufacturers in the world enter a momentous transition. Biocompatibility has emerged at the forefront of regulatory and commercial decision-making due to a rising interest in patient safety, material compatibility, and long-term device performance.
The Medical Device Event 2026 is one of the major medical device events of 2026 that addresses growing concerns regarding chemical characterisation, toxicological risk assessment and lifecycle safety. Manufacturers must be compliant with minimum standards as new materials, combination devices and digital health products enter the market.
Medical device forum 2026 provides a structure to understand evolving expectations and practical implementation strategies.
Documentation alone is not sufficient in today’s increasingly complicated regulatory environment. I believe knowledge sharing, peer learning, and early identification with regulatory developments are important.
The Medical Device Conference 2026 brings together internationally recognised MedTech leaders to share case studies, inspection learning, and compliance best practices. The Medical Device Forum 2026 provides for peer learning and strategic collaboration to provide for process benchmarking and quality improvement.
A leading international event on medical devices, the conference is a bridge between regulation, science and commercial success in 2026.


Regulatory Pressure: EU MDR vs FDA Expectations
Industry leaders attend forums not just for information but also to gain strategic advantage. The Medical Device Conference 2026 is an annual gathering of executives, regulators, R&D heads, and quality professionals that will gather for actionable insights.
As the premium MedTech forum 2026, it allows for direct access to regulators, notified bodies, testing laboratories, and solution providers. The Medical Device Forum 2026 also allows for meaningful discussions about future-ready compliance models, digital transformation, and sustainable innovation.
This medical device event 2026 is a must-see platform for companies looking to grow their competitiveness.

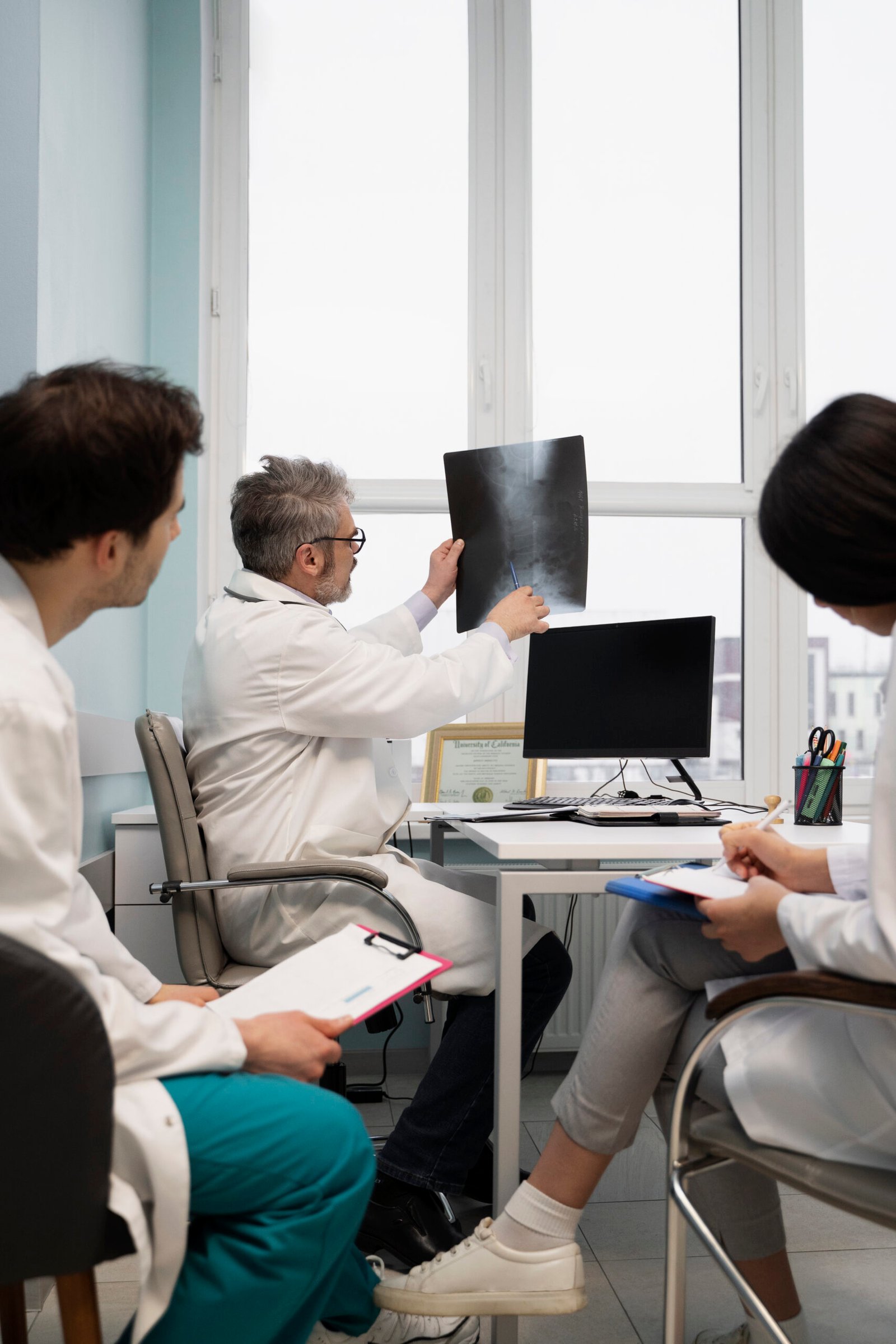
Monbien Events curates business-driven and intelligence-rich events that respond to real regulatory and technical problems. Not a little bit of a joke, the Medical Device Conference 2026 is no exception.
With over 16,000 attendees, it is the largest medical device event in 2026, bringing together expertise, case studies, and networking experiences on one global stage. This Medtech Forum 2026 has a focus on innovation, compliance, and growth, and members leave with knowledge that they can apply right away.
The Medical Device Conference 2026 brings together top MedTech leaders from around the globe, which makes it a must-see for anyone serious about regulatory success and patient safety in 2026 and beyond.

Europe hosts several top-rated medical device regulatory conferences, particularly those focused on EU MDR and IVDR, often integrated into larger B2B summits and compliance-focused forums.
Germany frequently hosts medical device biocompatibility conferences, workshops, and symposiums that focus on ISO 10993, material safety, and biological evaluation.
Yes, many organisers now offer hybrid and virtual medical device regulatory events, allowing global participation and flexible learning options.
Exhibiting at medical device safety conferences helps testing labs, consultants, and solution providers showcase expertise, connect with decision-makers, and generate qualified leads.
You can compare agendas, speakers, exhibitor profiles, and regulatory focus areas to determine which medical device regulatory conference best suits your professional or business goals.
Our clients share their experiences – honest feedback that reflects trust, impact, and satisfaction.








